|
Elements are either metals, nonmetals, or metalloids (or semi metals). The alkali metals (group 1) are very reactive, readily form ions with a charge of 1+ to form ionic compounds that are usually soluble in water, and react vigorously with water to form hydrogen gas and a basic. These elements are representative metals, metalloids, and nonmetals. Electricity and heat can travel through metalloids but not as easily as they travel through metals.\): Types of Elements. The representative elements occur in groups 1, 2, and 1218. They can be shiny or dull and their shape is easily changed. The shape of nonmetals cannot be changed easily because they are brittle and will break.Įlements that have properties of both metals and nonmetals are called metalloids. periodic tables), and a traditional dividing line between metals and nonmetals. As compared to metals, they have low density and will melt at low temperatures. Some periodic tables include a zig-zag line to distinguish between metals and metalloids. Their surface is dull and they don’t conduct heat and electricity. Using colored pencils, color each group on the table as follows. Yellow Boxes - metalloids Light Blue Boxes - nonmetals Dark Blue Symbols - Solids Magenta Symbols - Gases Bright Blue Symbols (Hg and Br) - Liquids. 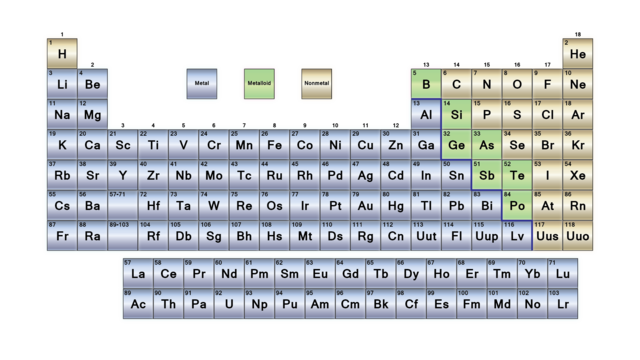
Nonmetals, on the right side of the periodic table, are very different from metals. 
Heat and electricity travel easily through metals, which is why it is not wise to stand next to a flagpole during a thunderstorm! Metals will corrode, gradually wearing away, like rusting iron. 
Nonmetals often have a drab look, low melting temperatures, boiling boils, and densities, and are poor heat and. Metals are excellent heat and electricity conductors, as well as malleable (able to be hammered into sheets) and ductile materials (can be drawn into wire). The line begins below boron (B) and extends between bismuth (Bi) and polonium (Po) or down between livermorium (Lv) and Tennessine (Ts). Answer: Elements are made up of metals, non-metals, and metalloids. Their shape can be easily changed into thin wires or sheets without breaking. Some periodic tables include a zig-zag line to distinguish between metals and metalloids. Conduction: They are poor conductors of heat and electricity. Non-Malleable and Ductile: Non-metals are very brittle, and cannot be rolled into wires or pounded into sheets. There are 118 elements known to us, out of which 92 are naturally occurring, while the rest have been prepared artificially. They are usually shiny, very dense, and only melt at high temperatures. Physical Properties of nonmetals: Physical State: Most of the non-metals exist in two of the three states of matter at room temperature: gases (oxygen) and solids (carbon). 
The periodic table on the left separates elements into three groups: the metals (green in the table), nonmetals (orange), and metalloids (blue). What's a molecule? Metals, Nonmetals, & Metalloids
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
